

The Lab


The main goal of Matias Simons Lab is to understand the molecular mechanisms underlying kidney disease.
Guided by “real-world” human genetic data, we are particularly interested in exploiting renoprotective gene variants for therapeutic strategies and in understanding how kidney cells maintain fitness in stress conditions. Our most recent focus in this context is on lipid metabolism and the regulation of membrane fluidity in proximal tubular cells.
The lab is part of the Institut für Humangenetik • of the Universitätsklinikum Heidelberg as well as the Molecular Medicine Partnership Unit (MMPU) with the European Molecular Biology Laboratory (EMBL).

Research
(Patho)physiology of proximal tubular cells of the kidney
Specialized epithelial cells constitute the dominant executors of tissue-specific physiological functions. In the kidney, our main organ of interest, there are a number of different epithelial cells organized in segments along the nephron, and we are particularly interested in the proximal tubules.
Epithelial cells of the proximal tubules have a very active endolysosomal system, and this is because its main task is to reabsorb virtually all the proteins that are filtered by the glomerulus. For this, the apical brush borders are equipped with a dedicated protein uptake pathway, involving the multiligand receptors megalin and cubilin. Failure of this pathway results in low-molecular-weight proteinuria, which is a hallmark of proximal tubulopathies (e.g. cystinosis and Dent’s disease).
Our recent research in Drosophila has introduced a novel mechanism for the control of apical protein uptake with strong implications for proximal tubular cells. Our findings propose that lysosomal mTOR signaling – a major nutrient sensing pathway that controls metabolic decisions from the lysosomal surface – regulates the expression of megalin as well as the morphogenesis of the apical surface. Therefore, we are studying how protein and lipid ligands from the tubular lumen can amplify a cycle of endocytosis and lysosome-to-nucleus signaling to satisfy the high metabolic needs of proximal tubular cells.
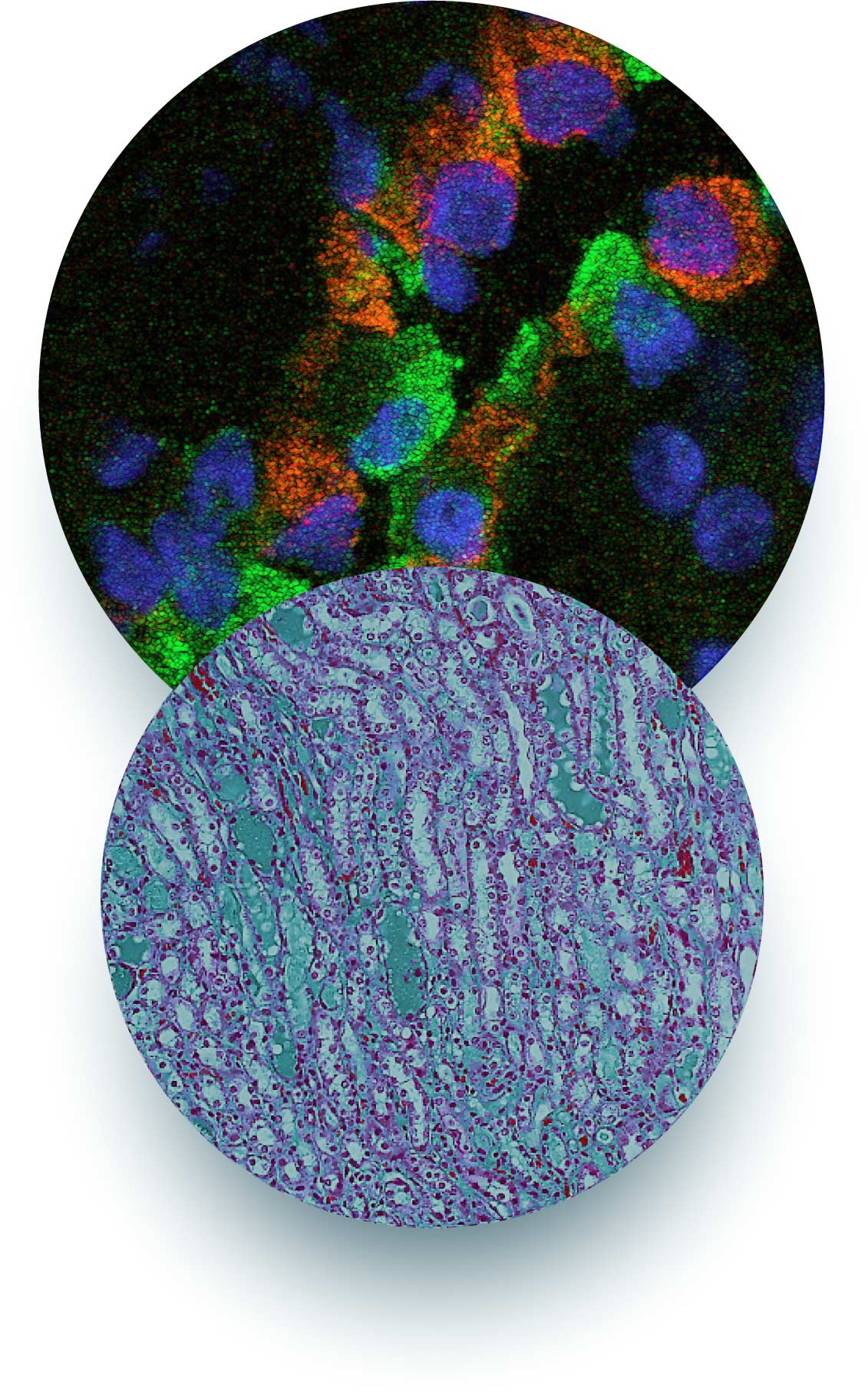
Targeting tubular albumin uptake and lipotoxicity as a renoprotective strategy
Glomerular injury can lead to an overload of the proximal tubules with proteins. Particularly damaging is the uptake of albumin, because albumin can carry toxic molecules, e.g. fatty acids, inside the cells.
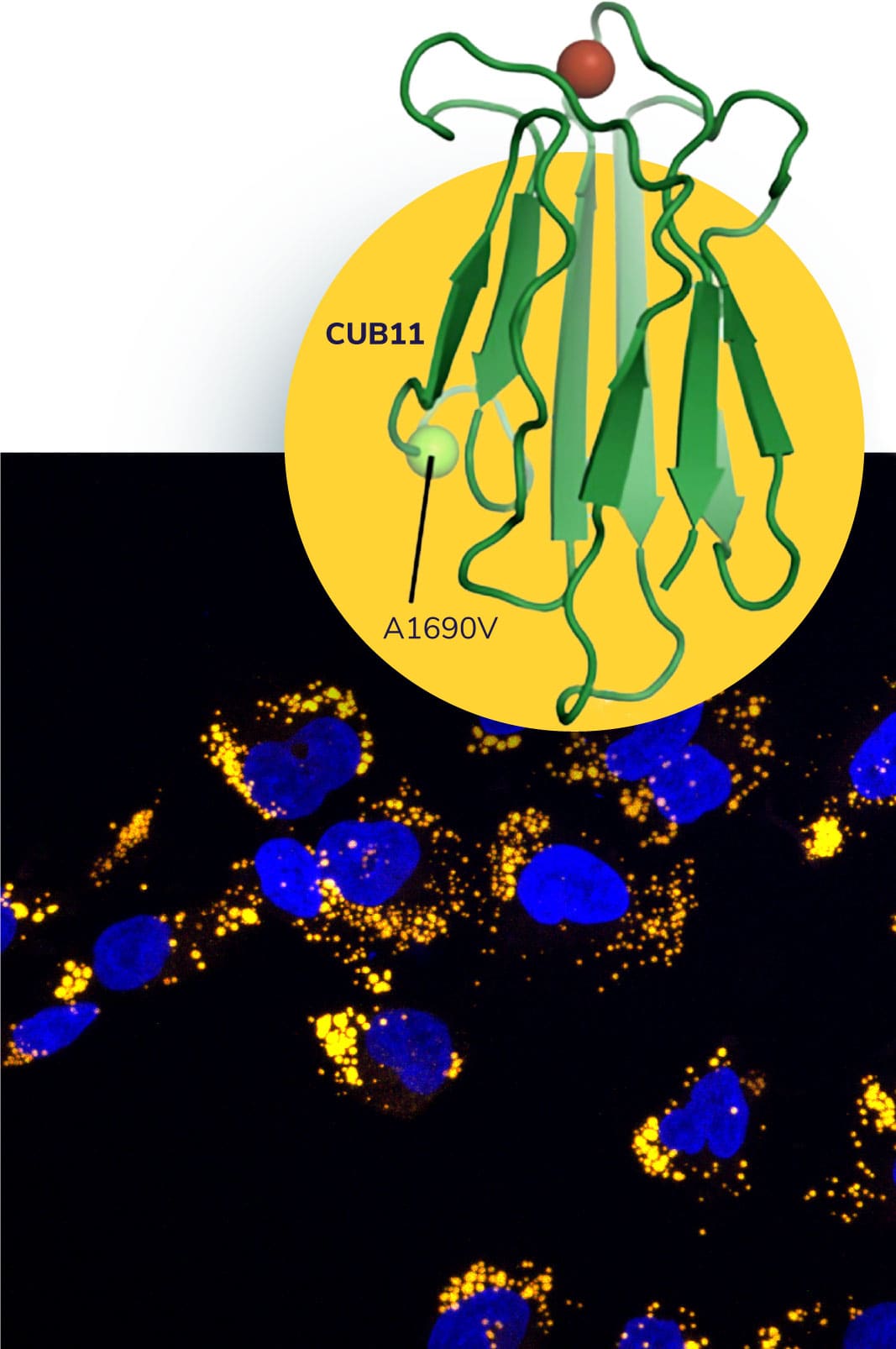
Our recent identification of patients carrying biallelic mutations of cubilin (gene name: CUBN) as well as population-based studes on more common CUBN variants suggest that dysfunctional cubilin leads to chronic proteinuria with normal renal function.
CUBN-associated proteinuria was featured by high percentage of albuminuria, suggesting that cubilin could indeed be the main albumin receptor in the proximal tubules. Based on these findings, we propose that the targeting of cubilin could be a safe strategy in conditions with glomerular proteinuria, such as diabetic kidney disease.
In addition, we found that saturated fatty acids can cause ER stress upon uptake via albumin. Mechanistically, ER stress is caused by elevated levels of saturated TAG precursors, reduced LD formation and, consequently, higher membrane order in the ER. Simultaneous addition of OA can rescue the cytotoxic effects by normalizing membrane order and by increasing both TAG and LD formation. As a future aim, we want to identify cellular pathways that can prevent lipotoxicity by enhancing lipid unsaturation.
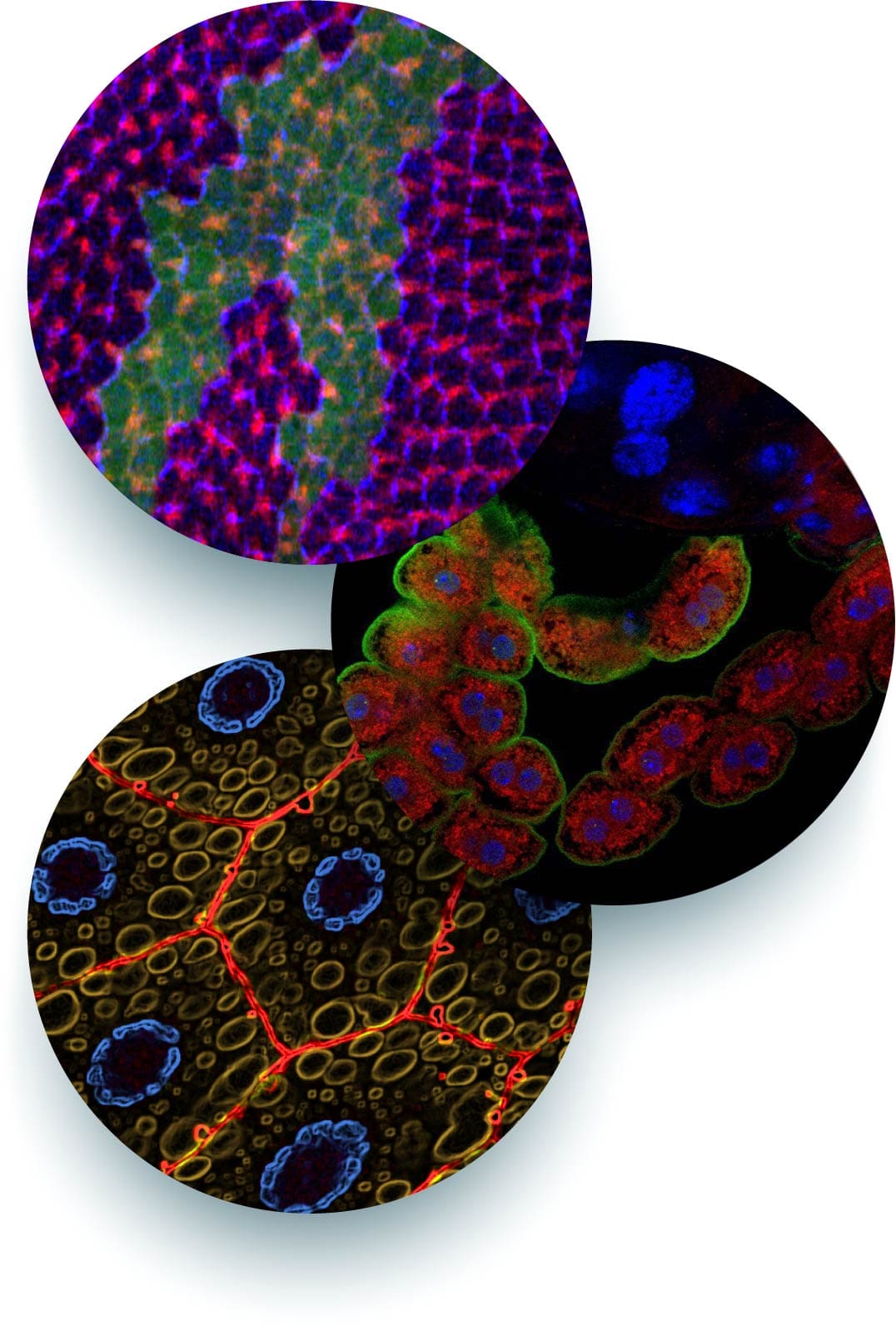
Drosophila as a tool in human genetics
The understanding of human genetic diseases has been greatly improved by novel techniques, such as next generation sequencing, allowing the complete genotyping of vast numbers of affected individuals and their relatives.
Moreover, novel genome editing methods and reprogramming of patient-derived cells have enhanced the possibilities for functional follow-up studies.
However, the evaluation of the pathogenicity of genetic variants remains a major bottleneck, because the human genome still lacks important functional gene information.
An important goal of the lab is to employ the Drosophila model as an innovative toolkit for the rapid identification of novel genes for hereditary diseases, particularly in the area of kidney disease but also other diseases.
Team
Questions? Interested in joining us?
Feel free to contact us.
 Franziska Meichsner Master Student
Franziska Meichsner Master Student  Simon Hammer MD Student
Simon Hammer MD Student
Publications
Explore our latest publications here













